Part 1 - Gas.
Gases boil at different temperatures. When a gas goes above it's 'boiling point' it will change state from a liquid to a vapour. Our stoves require a minimum pressure difference between the outside ambient air pressure and the pressure inside the canister so we get a good flow of gas to the burner. As a rough guide you want the gas temperature to be about 10C above the boiling point of that gas.
We typically use three different gases in New Zealand for outdoors stoves. They are n-Butane , Isobutane and Propane. Pure gases. Isobutane is the same atoms of n-butane rearranged which changes it's properties. Note: These are all Liquified Petroleum Gases. What we buy for BBQ's in New Zealand is typically a mix of Propane:Butane with a ratio of 60:40.
The boiling points of these gases at sea level are:
n-Butane: -1C
Isobutane:-11.7C
Propane:-42C
Pressure is a function of the volume of the container and the temperature of the gas. So, let's imagine we could buy three identical volume canisters from the store each containing one of these three gases. It's a real cold morning, -1C. We sit the gas canisters outside for an hour and they're now at -1C also. If the volume is the same and the temperature is the same they'll all be the same pressure right? No. The temperature of the gases is all the same at -1C but the temperature of the gases above their individual boiling points is different. -1C is a damn hot day to Propane. You try get your stove to light with that n-Butane canister and their is no pressure inside to push any gas out.
So why can't we buy Propane in those handy disposable canisters we all use for hunting if it's so great for cold temperatures? The canisters are manufactured to a European standard called EN417. EN417 outlines specifications for disposable gas canisters 50ml-1000ml. It states at 50C the maximum pressure must be no greater than 13.2bar. Propane reaches that around 40C at sea level and it's a steepening curve as can be seen in the below picture.
You can buy 100% propane however but it will be in a heavier duty container and you'll notice it will have stronger thicker walls, welded joins and a safety pressure relief valve.It will also have a different connection on it, probably a CGA600 connector.
Our little EN417 canisters are very thin and typically crimped at the top and bottom and we have what is called a Lindal valve for connection to our stoves. Below is an image of a canister I've opened up so we can see how it's made.
Yellow circle: Lindal valve
Green circles: Seals
Red circles: Crimps
Notice how the base of the canister is inverted? EN417 requires that base to overt before the canister ruptures. They do overt but in reality they often overt and milliseconds later the canister explodes so you wouldn't rely on it to overt and warn you in time it's going to explode soon.
What we are going to be buying for hunting is a mixture of Isobutane and Propane. The mix ratio will depend on the brand. MSR IsoPro gas for example is 80/20 Isobutane and Propane. It is an excellent cold weather gas with a boiling point of approximately -22C.
So, takeaways from this section : n-butane no good for colder temps - probably the cheapest gas at the store. Propane is high pressure but good for very cold temps but requires a different stove to connect to the bottle. Isobutane mixes are the best for our typical hunting trips where we are carrying our stoves and gas - different brands have different mixes.
Next I'll start showing you the different styles of stoves and some limitations of each type and some techniques to get around (to a point) these limitations.
Welcome guest, is this your first visit? Create Account now to join.
Welcome to the NZ Hunting and Shooting Forums.
Search Forums
User Tag List
Results 1 to 15 of 54
Thread: All about gas stoves.
Threaded View
-
12-04-2020, 11:14 AM #1
All about gas stoves.
Similar Threads
-
Vargo Ti Triad/Triad Multi-Fuel Stoves
By Nesika in forum Gear and EquipmentReplies: 8Last Post: 10-03-2016, 10:56 PM
Tags for this Thread
Welcome to NZ Hunting and Shooting Forums! We see you're new here, or arn't logged in. Create an account, and Login for full access including our FREE BUY and SELL section Register NOW!!




 67Likes
67Likes LinkBack URL
LinkBack URL About LinkBacks
About LinkBacks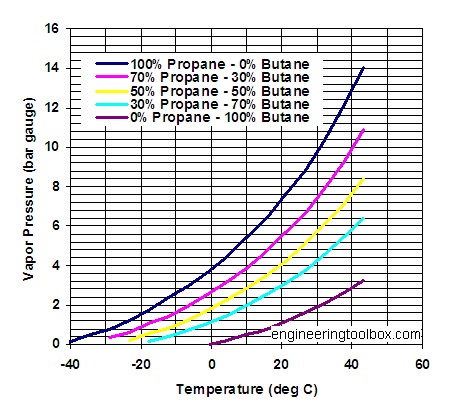




 Reply With Quote
Reply With Quote


Bookmarks